Lesson Plans
Modeling Salinity and Deep Ocean Currents
Overview
In this experiment, students make a claim about the cause of ocean currents and then develop a model to explain the role of salinity and density in deep ocean currents. This lesson is modified from "Visit to an Ocean Planet" Caltech and NASA/Jet Propulsion Laboratory.
Materials Required
- 4 Baby food jars
- 2 Laminated index cards
- ½ cup Table salt
- 2 Colors of food coloring (red and blue)
- 1 Stir stick
- Dishpan (for spills)
- Towels
- Map of surface currents
- Map of surface salinities
- Optional: aprons
- Student Data Sheet
PREPARATION
- It is important to do this activity before your students do it. This will give you a chance to see and work out any potential problems beforehand. Gather the supplies or send a supply list home with the students. Make sure that the students mark their names on anything they bring to class that will be returned home.
Procedure
Engage the Learner:
-
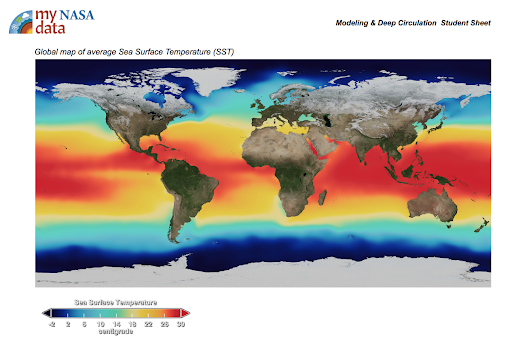
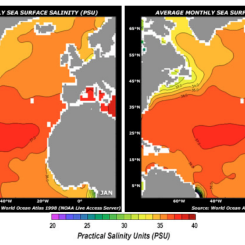
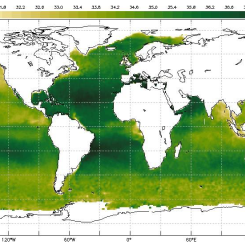
Display the maps of surface ocean currents and surface salinities of the oceans [Figs. 1 & 2].
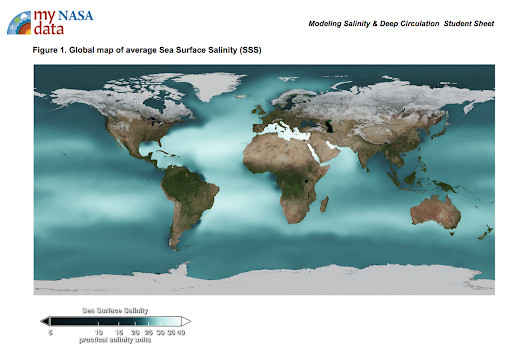
Figure 1. Sea Surface Salinity 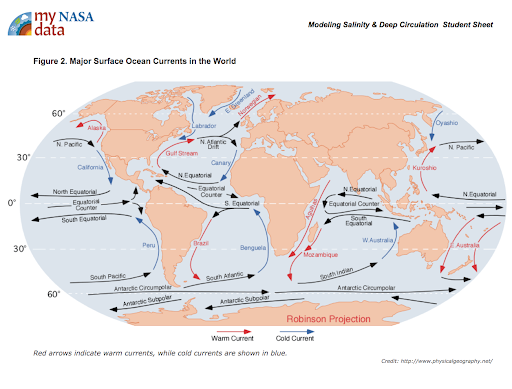
Figure 2. Surface Ocean Currents -
Describe the color bar; it shows the long-term
 average sea surface temperature, with dark green depicting less saline waters and white depicting more saline waters.
average sea surface temperature, with dark green depicting less saline waters and white depicting more saline waters. -
Have the students look for relationships between salinity and the locations of warm and cold currents.
-
Distribute the Student Sheet. Direct students to write their observations down on Student Sheet in the Initial (Quantitative/Qualitative) Observations section.
Asking Questions:
- Next, have students brainstorm questions that come to mind about these data and how they may be related. Students write their questions in the Your Questions section.
- Guide students through the process of generating a claim statement. Ask the students to write a claim that addresses these relationships in the Claim Statement section. Example., Surface currents control the water’s salinity.
Carrying Out the Investigation:
First model the experiment for the students before having them conduct the investigation with their group:
- Fill both baby food jars with water of the same temperature. Note to students the parts of the experiment that you are holding constant: jars, amount of water, the temperature of the water, etc.
- Dissolve ¼ salt in one of the jars and add blue food coloring. Make sure to mark the jar “Salt Water.”
- Add three drops of red food coloring to the other jar and label it “Fresh Water.”
- Place a 3 x 5 index card (laminated, preferred) on top of the saltwater and carefully & quickly invert it. Place the saltwater jar on top of the freshwater container and have someone carefully remove the card.
- Observe the results.
- Use the second set of jars to repeat the experiment.
- This time, invert the freshwater jar over the saltwater jar. Remove the card, and observe the results.
- Take both sets of jars, turn horizontally, remove the card and observe the results.
Analyzing the Results:
- Allow time for students to document their observations in the Evidence section of the dataset.
- Students observations should include the following:
- When the saltwater is placed on top of the freshwater, the water in the two jars will mix and the saltwater will sink to the bottom.
- When the freshwater is placed on top of the saltwater, the freshwater will sit on top of the salty water and no mixing will occur.
Interpreting Data:
Provide the following questions to help students with their observations/evidence:
- Describe how the salt and freshwater moved in the experiment? Saline water sunk to the bottom, under fresh water.
- Why does the salty water sink? It is denser (has more molecules in the same amount of space).
Explain The Results:
Facilitate a discussion based on the following points:
- The greater the amount of salt in the water, the higher its density will be because there are more molecules found in the jar. The saltwater will be heavier than the freshwater so when salt water is placed on top of freshwater the two will mix and the saltwater will sink to the bottom. When freshwater is placed on top of saltwater the higher density water is already on the bottom and is held there by gravity so no mixing occurs.
- This model displays the movement of water based on its salinity, especially in the deep ocean. When salty water sinks deeper through the water column because of its greater density, deep ocean currents are formed as the water moves horizontally and travels from areas of high salinity to those that are less salty. The salinity of the water may increase due to evaporation or freezing of water into sea ice at the poles.
- Queue the Sea Surface Temperature animation. Explain the color bar used in the following animation shows the long term average sea surface salinity, where white regions have the highest salinity and dark regions the lowest.
- Students will observe the video and look for locations that show patterns of higher salinity regions vs low.
- Run the video of Sea Surface Salinity.
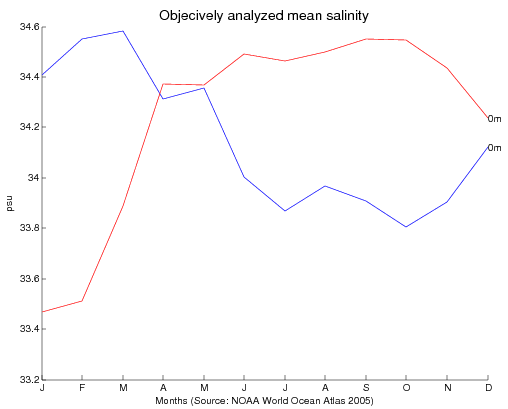
- Discuss the key findings. Students should observe the higher salinity values of the Atlantic than the Pacific. This is due to the greater rainfall amounts in the Pacific, and the lower salinity at the mouths of major rivers.
-
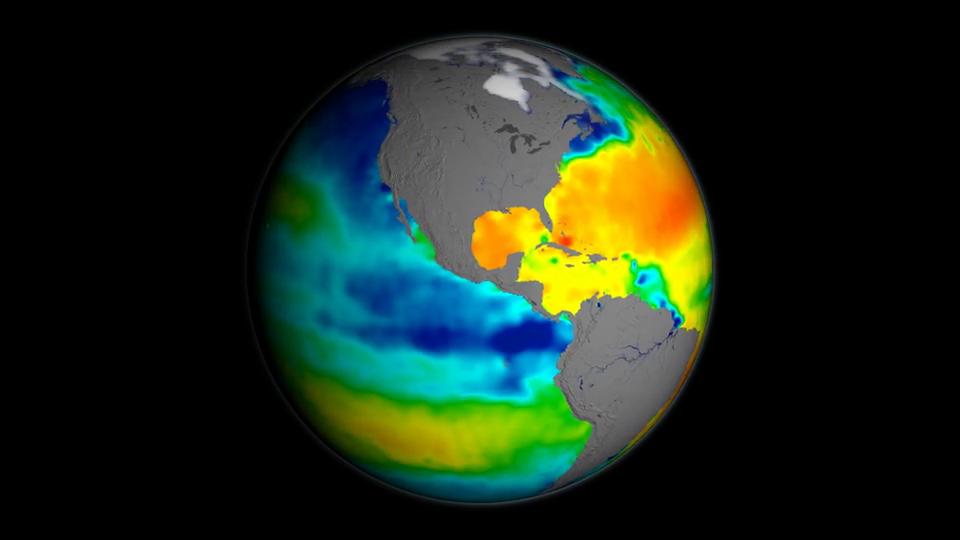
Display the following image, drawing students' attention to the Atlantic Ocean basin.
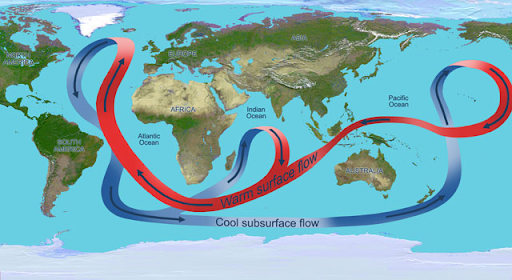
Thermohaline Circulation Credit: NASA - Ask what is happening here? The Atlantic features a circulation pattern of warm surface currents, including the Gulf Stream, that brings warm surface waters from the tropics northward into the North Atlantic. There, in the seas surrounding Greenland, the water cools; sea ice forms removing freshwater from the water making it denser, sinking to great depths and changes direction. What was once warm surface water heading north turns into cold deep water going south. This overturning is one part of the vast conveyor belt of ocean currents that move heat around the globe.
- Present the following question to the students: What effect may increasing air temperatures have on this conveyor belt that distributes heat around the world? Increasing air temperatures will cause an increase in the melting of land and sea ice in the polar regions. This would release more freshwater into the polar regions and may affect the salinity-driven deep water circulation.
Constructing Explanations:
Engage students in a discussion around the following questions/tasks:
- What water is heavier: salt or freshwater? Saltwater
- Which has the greatest density (the amount of “stuff” in a given amount of space)? Explain. Saltwater has a greater density because it has a greater amount of “stuff” (mass) in its volume. The saltwater has the water plus the dissolved salt, while freshwater does not contain the extra “stuff” (mass) of the dissolved salt.
- Where do you expect to find the most saline ocean water? Deep ocean
- What may explain why the surface waters at the poles have lower salinity values? The more saline waters sink to the deep ocean.
- What explains why the surface salinity values (Figure 2.) are so high at the equator and mid latitudes? Students may say that the water is warmer; the sun’s radiation is more intense at the equator; there is greater evaporation in this area.
- Now have students return to their student sheet and write the scientific principles that affect the movement of water based on salinity. They should connect scientific understanding to their observations in the Evidence section.
- How does our understanding of the science processes affecting the movement of water in the oceans based on their salinity values relate to their Claim Statement.
- Is your Claim supported by your evidence and backed by our scientific understanding of the ocean circulation? Why or Why Not?
- Have students share their Claim Statements with the class and how their deeper dive into salinity-driven currents have changed their understanding of the Hydrosphere.
The Student will:
- Develop a claim about the cause of ocean currents
- Model to explain the connection between salinity and density in ocean currents.
- How do ocean properties affect the movement of water?
The oceans are mostly composed of warm salty water near the surface over cold, less salty water in the ocean depths. These two regions don't mix except in certain special areas. The ocean currents, the movement of the ocean in the surface layer, are driven mostly by the wind. In certain areas near the polar oceans, the colder surface water also gets saltier due to evaporation or sea ice formation. In these regions, the surface water becomes dense enough to sink to the ocean depths. This pumping of surface water into the deep ocean forces the deep water to move horizontally until it can find an area on the world where it can rise back to the surface and close the current loop. This usually occurs in the equatorial ocean, mostly in the Pacific and Indian Oceans. This very large, slow current is called the thermohaline circulation because it is caused by temperature and salinity (haline) variations.
This animation shows one of the major regions where this pumping occurs, the North Atlantic Ocean around Greenland, Iceland, and the North Sea. The surface ocean current brings new water to this region from the South Atlantic via the Gulf Stream and the water returns to the South Atlantic via the North Atlantic Deep Water current. The continual influx of warm water into the North Atlantic polar ocean keeps the regions around Iceland and southern Greenland mostly free of sea ice year-round.
The animation also shows another feature of the global ocean circulation: the Antarctic Circumpolar Current. The region around latitude 60 south is the only part of the Earth where the ocean can flow all the way around the world with no land in the way. As a result, both the surface and deep waters flow from west to east around Antarctica. This circumpolar motion links the world's oceans and allows the deep water circulation from the Atlantic to rise in the Indian and Pacific Oceans and the surface circulation to close with the northward flow in the Atlantic.
The color on the world's oceans at the beginning of this animation represents surface water density, with dark regions being most dense and light regions being least dense (see the animation Sea Surface Temperature, Salinity and Density). The depths of the oceans are highly exaggerated to better illustrate the differences between the surface flows and deep water flows. The actual flows in this model are based on current theories of the thermohaline circulation rather than actual data. The thermohaline circulation is a very slow moving current that can be difficult to distinguish from general ocean circulation. Therefore, it is difficult to measure or simulate.
- Ability to read maps
- Knowledge of safety procedures while conducting science experiments and models
- Basic understanding of density
- Earth’s oceans are separate individual bodies of water and are not connected.
Salt plays an important role in how the Earth system functions. It flows through Earth's ocean, the lifeblood of Earth's climate system. The ocean is roughly 3.5% salt and the concentration of dissolved salts in the ocean is referred to as salinity, which varies across the globe and over time. Just as too much or too little salt in our diets affects our health, so too do high and low salinity have profound effects on how the ocean circulates, how freshwater cycles around Earth and how our climate works. The concentration of salt on the ocean surface — the part of the ocean that actively exchanges water and heat with Earth's atmosphere — is a critical driver of ocean processes and climate variability. To better understand the regional and global processes that link variations in ocean salinity to changes in the global water cycle - and how these variations influence ocean circulation and climate - NASA built and launched Aquarius, the primary instrument aboard the international Aquarius/Satélite de Aplicaciones Científicas (SAC)-D observatory.
- Biological oceanographers and marine biologists study plants and animals in the marine environment. They are interested in the numbers of marine organisms and how these organisms develop, relate to one another, adapt to their environment, and interact with it. To accomplish their work, they may use field observations, computer models, or laboratory and field experiments.
- Chemical oceanographers and marine chemists study the composition of seawater, its processes and cycles, and the chemical interaction of seawater with the atmosphere and seafloor. Their work may include analysis of seawater components, the effects of pollutants, and the impacts of chemical processes on marine organisms. They may also use chemistry to understand how ocean currents move seawater around the globe and how the ocean affects climate or to identify potentially beneficial ocean resources such as natural products that can be used as medicines.
- Geological oceanographers and marine geologists explore the ocean floor and the processes that form its mountains, canyons, and valleys. Through sampling, they look at millions of years of history of sea-floor spreading, plate tectonics, and oceanic circulation and climates. They also examine volcanic processes, mantle circulation, hydrothermal circulation, magma genesis, and crustal formation. The results of their work help us understand the processes that created the ocean basins and the interactions between the ocean and the seafloor.
- Physical oceanographers study the physical conditions and physical processes within the ocean such as waves, currents, eddies, gyres and tides; the transport of sand on and off beaches; coastal erosion; and the interactions of the atmosphere and the ocean. They examine deep currents, the ocean-atmosphere relationship that influences weather and climate, the transmission of light and sound through water, and the ocean's interactions with its boundaries at the seafloor and the coast.
- Internet Required
- Teacher computer/projector only